Human Papillomavirus (HPV) is a virus that is transmitted via human contact and is the most common sexually transmitted infection in the UK and US. It can infect the skin, genitals and the oral and anal cavities. It is a very common virus; in fact it’s thought that about 80% of us will be infected with it at some stage in our lives. Luckily, many cases of HPV are eradicated by our own immune system without causing any problems or presenting with any symptoms.
We’ve known about HPV for a while now, particularly since the early 1980s when a significant role of HPV was found in the cause of cervical cancer. Nowadays, HPV is known as the leading cause of cervical cancer, with almost 99% of cervical cancers proving positive for HPV. Despite this strong correlation, it has also been found that, in the absence of any other signs, a HPV positive cervical sample does not necessarily indicate cancer.
In a recent study, published in the November 2011 issue of Journal of the American College of Cardiology, scientists from the University of Texas claim that “Human Papillomavirus infection, especially cancer-associated oncogenic types, is associated with cardiovascular disease (CVD) among women”.
Authors Kuo and Fujise are working on why almost 20% of CVD cases (heart attack or stroke) cannot be explained by the usual risk factors (cigarette smoking, high blood pressure, high blood cholesterol or diabetes). They believe that HPV may be partly culpable.
Presently, over 100 different types of HPV have been identified. These types are subdivided into two groups; those that have been found to cause cancer (high-risk HPV types) and those that cause genito-anal warts and do not cause cancer (low-risk HPV types). It is the high-risk, or “cancer-associated oncogenic” HPV types, that the authors are particularly interested in.
The study focuses on the main cause of heart attacks and strokes – atherosclerosis, which is the thickening and hardening of artery walls due to a build up of fat and inflammatory cells. The authors note that our bodies normally regulate the process of atherosclerosis with a protein we produce daily, called p53, which functions to promote the breakdown of the hardened fatty plaques and prevent further building up in the arteries [ref]. So, when the early signs of artery wall thickening appear, our bodies begin to produce more p53 to combat it.
The gene that codes for the p53 protein (TP53) is a well known tumour suppressor gene, essentially meaning that it functions to prevent the growth of tumours. If TP53 is defective or absent, the risk of that individual developing a tumour is increased (I’m sure you appreciate this is simplified for the purpose of this blogpost) and so it is not surprising that the most common genetic aberrations found in cancers are a mutation or deletion in the TP53 gene [ref]. When the function of p53 is lost, either by inhibition of the protein or problems with the TP53 gene, an atheromatous plaque develops; numbers of cells such as vascular smooth muscle cells increase (because the mechanism by which cell multiplication is controlled is now lost) and many more inflammatory cells migrate to the site [ref].
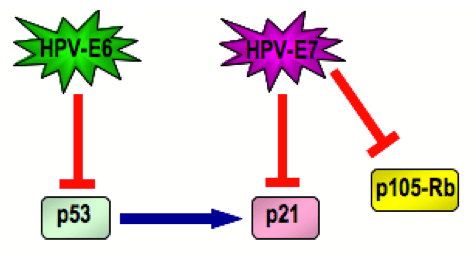
Coming back to the topic of HPV and its link with p53: when HPV infects a cell its genome becomes integrated with the host’s DNA and it does this in a manner of early and late stages. Early stages are denoted by the sequential expression of seven “early” (E) proteins, named E1-E7. The late stages express just two “late” (L) proteins, named L1 and L2. E6 and E7 proteins are both associated with cancer because they inhibit the products of two tumour suppressor genes, one of these being the p53 protein (inhibited by the HPV-E6 gene) and the other the retinoblastoma nuclear phosphoprotein, p105-Rb (inhibited by HPV-E7 gene). HPV-E7 also inhibits another protein, p21, which is involved in cell cycle progression but mostly under the control of p53.
Kuo and Fujise echo the well-documented role of HPV in anogenital cancers via the p53 inhibition mechanism (illustrated left). Given that the absence of p53 has also been identified as an accelerator of atherosclerosis [ref], they postulate that the p53 inhibiting nature of HPV may also be associated with CVD.
To investigate their hypothesis, the authors sourced their data from the National Health and Nutrition Examination Survey (2003-2006), which combines a series of questionnaires and physical examinations. 2450 women, aged 20-59, qualified for the final analysis. They answered questions based on their sexual, drinking, CVD and comorbidity history. Assessments were made on their CVD risk (obesity, smoking, diabetes, hypertension, cholesterol) and the participants also provided self-collected vaginal swabs. The swabs were used to test for the presence of 37 HPV types, including both high- and low-risk. CVD was confirmed if a heart attack or stroke had been previously diagnosed by a health professional.
Their results indicate a low prevalence of CVD; with just 2.5% (60/2450) of women self-reporting a diagnosis of either heart attack or stroke. Of these 60 women, 35% (21/60) were HPV negative, 31.6% (19/60) were low-risk HPV positive and 33.3% (20/60) were high-risk HPV positive. In the entire study sample, 46.6% (1141/2450) tested positive for HPV; 23% (573/2450) of which were positive for cancer-associated HPV types. Of the 573 women with cancer-associated HPV types, just 20 (3.5%) had CVD. Of the 568 women with non-cancer-associated HPV types, a similar proportion of 19 (3.35%) women had a self-reported diagnosis of CVD. Adjustments were made to take into account the variables associated with the individual’s race, sexual behaviour and CVD risk factor management. Accommodating these adjustments, the study suggests that cancer associated HPV infections have a “strong and independent correlate for CVD” and all other HPV infections have no association with CVD at all.
The authors discuss that their findings not only mean that HPV infection status may have a role in the early diagnosis of potential CVD risk factors, as well as the management and/or prevention of CVD in women, they also suggest the potential of the HPV vaccination to have cardiovascular benefits. Additionally, their conclusion stating that cancer-associated HPV types are more likely to be associated with CVD than other HPV types, seems a little attenuated given that only 20 women with CVD tested positive for high-risk HPV in total. To extrapolate such a bold conclusion from such a small sample size – and prior to omitting those with CVD risk factors – seems a little ambitious.
The CVD risk factors studied, however, were only those that increased the risk of atherosclerosis. Atherosclerosis is not the sole cause of CVD. Underlying genetic predispositions are important causes of CVD, but were overlooked in this study. When researching the possible explanation for a relatively small percentage (20%) of inexplicable causes of CVD, it is crucial to account for other known factors such as heredity and genetic predispositions. Genetic arrhythmia disorders such as Long QT Syndrome, hypertrophic cardiomyopathy and Wolff-Parkinson-White Syndrome do not form atheromatous plaques but can lead to heart attacks through weak or loss of pumping. These conditions can also go easily undiagnosed and are thought to be responsible for approximately 25% of Sudden Cardiac Death incidences. Arrhythmia would not be detected by measuring variables only attributed to atherosclerosis, as carried out in this study. Similarly, there are possible genetic predispositions responsible for strokes, the main one being a mutation in the Notch3 gene, found in CADASIL (cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencepalopathy). Other genetic links to stroke have been discussed in this short review article [pdf]. Another factor not considered in this study when assessing CVD risk, was the individual’s predisposition to developing a blood clot – another important cause of heart attacks and strokes.
Age is a major risk factor for CVD [ref]; heart attacks mostly occur in people who are over 45 [ref] and strokes mostly occur in people who are over 65 [ref]. The sample age range, therefore, slightly reduces the likelihood of CVD cases being attributed to the individual’s age, but this is not definitive. Their results, however, only demonstrate the relationship between HPV infection status and three age groups (20-29, 30-45 and 46-59). Although they tabulated the results of the adjustments, including age, it would have been useful to see the direct relationship between age and CVD to ascertain if there are any skews towards a certain age group, particularly the ages 46-59, which are considered to be at greater risk of heart attack.
Despite the concerns from the authors about the potential problems with self-collected swabs, it is in fact well documented that there is actually an increased benefit of self-sampled cervical swabs for the detection of HPV [ref, ref, ref, ref], so results achieved by this method can be assumed reliable and accurate. The self-reported diagnosis of CVD, however, may not be as reliable. The one issue with the HPV test, however, is that it only detects the presence of HPV and does not determine how recent the infection occurred. This is a limitation of the study because the course of HPV infection may be important to the link with CVD, particularly if the low-risk HPV types can cause CVD but only after a longer course than high-risk HPV, perhaps.
The study also does not look into the relationship between HPV and CVD in men, however, the increased interest in the detection of HPV positive oral lesions [ref] may provide some valuable data for ascertaining whether or not there is a significance between HPV and CVD in men. This isn’t the first time an association has been made between CVD and an oral infection; In 2005, for example, Desvarieux et al described a direct link between heart disease and oral bacteria. The causal link between distant infections and CVD is generally thought to be related to the inflammatory cells that build-up around the infection that enter the bloodstream and become attached to the arteries or tissue in the heart. This is interesting because it implies a second mechanism for HPV to potentially cause CVD in women and now also one mechanism in men, not to mention suggesting that other infections have the potential to cause CVD also.
Given what is already known about p53 and its role in atherosclerosis, together with the effect of HPV on p53, the hypothesis that HPV may cause CVD does seem attainable, it’s the evidence that supports this claim that is not so easily producible. The authors have stressed that it would require more follow-up studies, collecting data about CVD that is more objective, as well as determining the exact mechanism by which HPV causes CVD, as this was not confirmed in this study. Perhaps, as HPV-E6 and HPV-E7 are most highly expressed in high-grade malignancies, it would have been useful to correlate the participant’s cervical screening Pap results also. Their study also failed to clarify if the HPV infection occurred prior to the diagnosis of CVD, and as correlation does not necessarily imply causation, it is vital for future studies to ascertain the chronology of these events. So while the link between HPV and CVD may seem theoretically likely, the causes of CVD are multifactorial and difficult to confidently denounce a single cause using methods as employed in this study and so a lot more work needs to be done before their claim can be accepted.
Discussion
No comments yet.